We are routinely told by surgeon thought leaders, implant manufacturers and sales reps that “robotic assisted surgery is here to stay” and that the robots will improve outcomes and reduce the often quoted and outdated “20% of knee replacement patients are not satisfied”. The slides are elegant. The data is cherry-picked. And the price tag is significant.
Will robotic-assistance improve outcomes in knee replacement over traditional optical navigation? Is there something about the actual mechanized arm of a robot that is better than a surgeon’s highly skilled arm if computer navigation determines the correct implant location?
That question has haunted the robotic surgery conversation in orthopedics for over a decade. Compared to manual instruments with no navigation? Sure, robotics wins on alignment precision. But compared to computer-assisted navigation - the technology that’s been available for nearly 30 years and costs a fraction of the price? Nobody had run that trial.
Until now.
The Study the Industry Didn’t Want
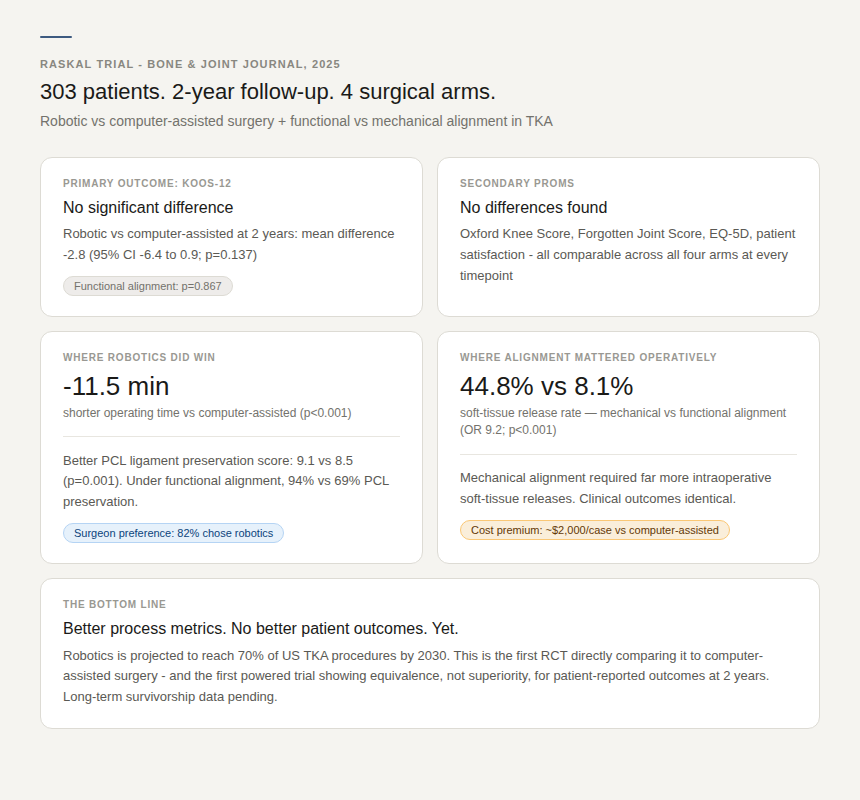
Published this year in The Bone & Joint Journal, the RASKAL trial (Robotic-Assisted Surgery and Kinematic ALignment in TKA) is the first properly powered randomized controlled trial to pit robotic-assisted surgery directly against computer-assisted surgery in total knee replacement.
303 patients. 14 surgeons across 11 hospitals. A 2x2 factorial design also testing functional alignment against mechanical alignment. Two-year follow-up with validated patient-reported outcomes.
The primary outcome: KOOS-12 - a 12-item patient-reported score measuring pain, function, and quality of life.
The finding: no significant difference.
Robotic versus computer-assisted surgery at two years showed a mean KOOS-12 difference of -2.8 points (95% CI -6.4 to 0.9; p=0.137). Functional alignment versus mechanical alignment: essentially identical (mean difference 0.3; p=0.867).
No differences in Oxford Knee Score, Forgotten Joint Score, patient satisfaction, or quality of life at any timepoint from three months to two years.
The system that healthcare’s been investing billions in - the one projected to be used in 70% of knee replacements in the United States by 2030 - produced outcomes statistically indistinguishable from a technology we’ve had since the 1990s.
Read that again.
What the Data Actually Shows - and What It Doesn’t
Before I go further, I need to be precise here. This is not a study saying robots are useless. I am not saying that. The data has nuance that the headline can’t hold.
Robotics did produce meaningful process improvements. Operating time was 11.5 minutes shorter with the robotic arm (p<0.001). PCL ligament preservation was measurably better - and under functional alignment, the robotic group showed 94% PCL integrity versus 69% with computer-assisted surgery.
Surgeons preferred robotics in 82% of cases. Functional alignment dramatically reduced the need for intraoperative soft-tissue releases - 8.1% versus 44.8% with mechanical alignment (OR 9.2; p<0.001).
These are real advantages. Shorter operative time is real. Ligament preservation is real. Reduced soft-tissue releases are real. But the decreased soft tissue releases is not due to the robotic arm…modern navigation systems are capable of enabling functional alignment.
What is not demonstrated - not yet, at two years - is that any of this translates into better outcomes for patients. Better technical execution did not produce better clinical results. That gap is something the field needs to sit with, honestly, rather than explain away.
Two important caveats. First, this trial was conducted in Australia using a specific robotic platform (Stryker Mako) and a single implant system (Triathlon). Results may not generalize to all robotic platforms. Second, two years is not the whole story. Where robotics may ultimately prove its value is in long-term implant survivorship - reduced aseptic loosening, lower revision rates. A 2025 retrospective analysis of 22,000 TKAs suggested exactly that. The RASKAL trial isn’t designed to answer the survivorship question. We need those answers.
But we should not let the long-term hypothesis excuse the short-term reality.
The $2,000-Per-Case Question
Here is the uncomfortable math.
The additional cost of robotic-assisted TKA has been estimated at approximately $2,000 per case versus computer-assisted surgery - and roughly $2,600 per case compared to manual instrumentation. In the United States, with roughly 700,000 knee replacements performed annually and robotic adoption climbing toward that projected 70% threshold, we are talking about hundreds of millions of dollars in additional healthcare spending per year.
For outcomes, at two years, that are statistically equivalent.
I am not anti-technology. My engineering degree sits next to my medical degree for a reason. I have been involved in implant design. I believe precision matters and that technology, properly applied, makes surgeons better. What I am against is the uncritical adoption of expensive technology driven by marketing momentum rather than clinical evidence - particularly in a healthcare system already collapsing under its own cost burden.
When I built Indiana Orthopedic Institute, one of the foundational principles was this: every dollar spent on care delivery must earn its place at the table. We have grown to over $40M in revenue and 100 employees, including 16 surgeons, in three years. We did that by asking hard questions about what actually improves patient outcomes - not what looks impressive in a brochure.
The question “compared to what” applies just as much to your capital budget as it does to your surgical approach.
The Moment I Got Humbled
I want to be honest about something.
I have used robotic assistance. I have felt the precision it offers - the way a well-planned robotic case tightens your confidence at the critical moments. There is a real subjective experience of control that the technology provides, and I understand why 82% of the surgeons in this trial preferred it.
But I have also had the harder experience: a patient - a 64-year-old retired teacher, otherwise healthy, excellent bone stock, a technically well-executed case by every objective metric - who at one year still had persistent stiffness and functional limitations that defied my surgical explanation. By all the numbers I track, that case was a success. She did not experience it that way.
What did I do differently for her? Not much, initially. And that failure to ask better questions earlier in her recovery is something I carry.
The point is this: technical precision and patient outcome are not the same variable. The RASKAL trial just proved that at scale. I proved it to myself one patient at a time.
That gap - between what we measure in the OR and what the patient actually lives - is where the real research frontier sits.
A Framework for Evaluating Surgical Technology
After 10,000+ joint replacements, I have developed what I call the Evidence-Before-Investment Framework for evaluating new surgical technology. It is not complicated, but it is disciplined.
1. Define the comparator. “Better than nothing” is not a standard. What is the best current alternative, and what is the data against that specific alternative?
2. Separate process metrics from patient outcomes. Alignment precision, operative time, and ligament preservation are process metrics. KOOS scores, Oxford scores, revision rates, and patient satisfaction are outcomes. Technology must eventually move the outcomes needle to justify its cost.
3. Understand the time horizon. Short-term equivalence may coexist with long-term superiority. Be honest about which claim the data supports at any given moment.
4. Account for cost in the value calculation. A technology that produces identical two-year outcomes at $2,000 more per case is not neutral - it is a net negative until proven otherwise. Our patients are already financially strained by the cost of care. That is not an abstraction; it is a moral responsibility.
5. Follow surgeon preference with skepticism. In the RASKAL trial, 82% of surgeons preferred robotics - even though outcomes were equivalent. Preference and efficacy are not the same. We are not immune to the cognitive biases that come with expensive, impressive tools.
What This Means for the Field
I want to speak directly to the surgeons and orthopedic leaders reading this.
The RASKAL trial is not the last word. It is the first rigorous word. What it demands is that we stop treating robotic adoption as inevitable progress and start treating it as a hypothesis that requires ongoing validation.
The technology may yet prove its value in long-term survivorship. The process improvements - shorter OR time, better ligament preservation, reduced soft-tissue releases - are clinically meaningful and may prove cost-effective when properly analyzed. I am not closing the door.
But the door should not have been this wide open without this data existing first.
The hospital systems and device companies that are pushing robotic adoption are not doing so primarily because of your patients’ two-year outcomes. They are doing so because of market share, capital investment recovery, and competitive positioning. That is not cynicism - it is just business. The question is whether you let their business logic drive your clinical decision-making.
Build your own framework. Ask “compared to what.” Read the trials, including the ones that challenge your existing investments.
Your patients deserve decisions made on evidence, not on the quality of the sales presentation.
That is the incision point. Where intention meets reality. Where the question “does this actually work better” has to be answered before the robot enters the room.
The RASKAL trial was published in The Bone & Joint Journal (MacDessi et al., 2025). Full citation: https://boneandjoint.org.uk/Article/10.1302/0301-620X.108B.BJJ-2025-1472.R2